View
comments
Remdesivir, the first drug to receive U.S. Food and Drug Administration (FDA) approval for treatment of COVID-19 patients may not improve mortality rates, a new study finds.
A research team led by the University of Iowa used data from the Veterans Health Administration to observe length of hospital stays and treatment outcomes among COVID-19 patients who were admitted to the hospital.
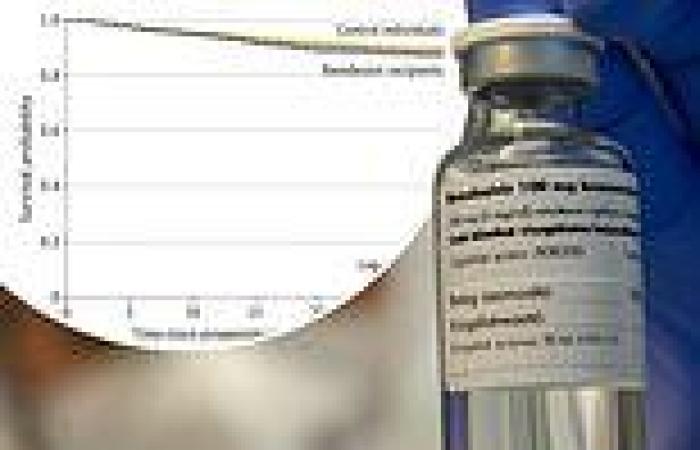
Results showed that there were no difference in mortality rates between those treated with remdesivir and those receiving standard care.
Patients who are treated with the drug have longer hospital stays as well.
Their findings show that the drug that once was the only one available for the condition may not be very effective at all.

There was little difference found in mortality rates between COVID-19 patients who received remdesivir and patients in a control group, a new study finds
The drug first received emergency use authorization from the U.S. Food and Drug Administration in May and received full approval in October 2020.
In the time since, three monoclonal antibody drugs have also been approved for emergency use treating the virus.
However, questions have since circulated about whether or not