View
comments
Millions of Americans rolled up their sleeves after the U.S. Food and Drug Administration (FDA) authorized COVID-19 vaccine booster shots.
Third shots of the Pfizer-BioNTech vaccine were given emergency use authorization on September 22 for those aged 65 or older, or 18 and older who are high risk of severe complications from the virus due to underlying conditions or their jobs.
Between September 22 and September 29, the White House reported that almost two million people received boosters.
The shots were in high demand after the White House announced plans to roll out boosters by September 20, though regulatory barriers prevented the shots from becoming available until a few days later.

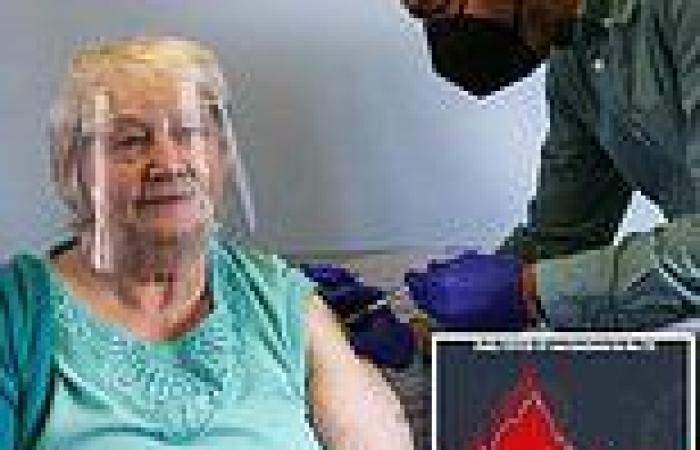
Almost two million Americans received Covid boosters last week, according to the White House. More than five million boosters have been distributed overall with a majority going to people over the age of 65. Pictured: an elderly woman in London, England, receives a shot of the Pfizer Covid vaccine on March 10
Jeff Zients, the White House COVID-19 response coordinator, announced the marker during a news briefing on Friday.
'As we vaccinate the unvaccinated, we're also