View
comments
A new antiviral COVID-19 pill developed by Merck & Co can reduce the risk of death from the virus a new study finds.
Molnupiravir was developed by the Kenilworth, New Jersey, based pharmaceutical company, can reduce risk of hospitalization or death by 30 percent.
The company is currently seeking emergency use authorization from the U.S. Food and Drug Administration (FDA), which will be discussed by agency advisors next week.
If the drug, which was developed in partnership with Ridgeback Biotherapeutics, receives approval in the U.S. - which it already has done in the UK - it will be the first oral medication approved to treat the virus.
A new study finds that molnupiravir (pictured), an antiviral pill developed by Merck, cut cut the risk of hospitalization or death from the virus by 30%
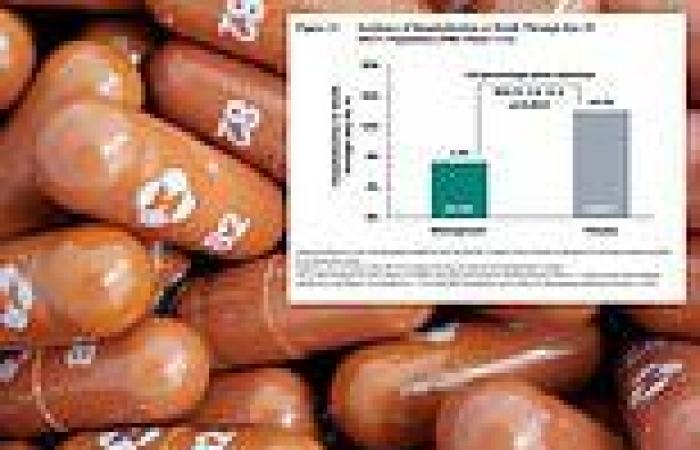
The study, published Friday by the FDA, included over 1,400 participants.
About 700 participants who had recently began to feel symptoms of Covid were placed in one of two groups.
One group received the drug, the other was given a placebo and operated as a control group.
Researchers found that 9.7 percent of the control group suffered a severe enough case to require hospitalization or cause death - compared